Hospital core labs
Automated chemistry, immunoassay, molecular, and sample handling workflows for high-volume test menus.
From high-throughput IVD analyzers to patient monitoring data flows, Roche Diagnostics helps clinical teams move results faster through LIS, HL7 FHIR, quality control, and post-market surveillance workflows.

Each program combines analyzer operations, result interoperability, connected service, and clinical governance in a sequence that laboratory directors can validate.
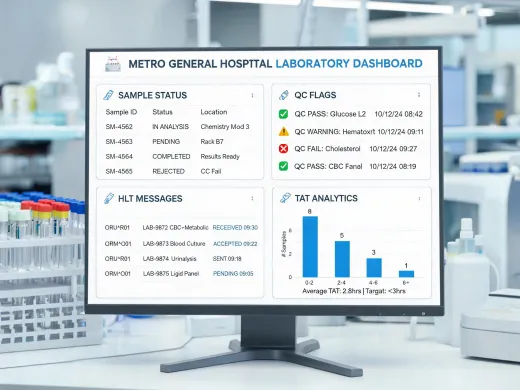
Bidirectional ASTM, HL7 v2.5.1, and FHIR Lab IG workflows keep orders, results, quality flags, and reflex testing logic synchronized across central labs, satellite labs, and point-of-care programs.

Remote telemetry supports preventive maintenance triggers, SBOM-aware patch planning, coordinated disclosure response, and service escalation before a queue delay becomes a patient care delay.

Linearity, precision under CLSI EP05, reportable range, reference interval, and method comparison evidence can be organized into value analysis packs for laboratory, procurement, and regulatory reviewers.
| Parameter | Central Laboratory | Connected Point of Care |
|---|---|---|
| Primary objective | High-throughput test consolidation with automated QC review. | Near-patient result delivery with governed remote oversight. |
| Integration layer | LIS middleware, bidirectional HL7 v2.5.1, reflex rules, QC dashboards. | FHIR observation push, device fleet enrollment, user role governance. |
| Validation evidence | Precision, linearity, Deming regression, Bland-Altman bias, CAP PT records. | Operator competency, connectivity audit, lot traceability, result review workflow. |
| Service model | Predictive analyzer maintenance, reagent cold-chain trace, uptime SLA. | Fleet firmware orchestration, cybersecurity bulletin review, remote support triage. |
Automated chemistry, immunoassay, molecular, and sample handling workflows for high-volume test menus.
Extended validation packs, reagent economics, lot traceability, and middleware control for distributed clients.
Point-of-care result routing, operator lockout rules, and critical value communication paths.
Targeted diagnostics and monitoring programs for oncology, cardiometabolic care, and infectious disease pathways.
Patient monitoring signals, adherence views, and follow-up workflows that do not expose identifiable patient imagery.






Bring Roche Diagnostics into analyzer selection, LIS architecture, service coverage, and remote care governance before the validation calendar is locked.
Request a Demo